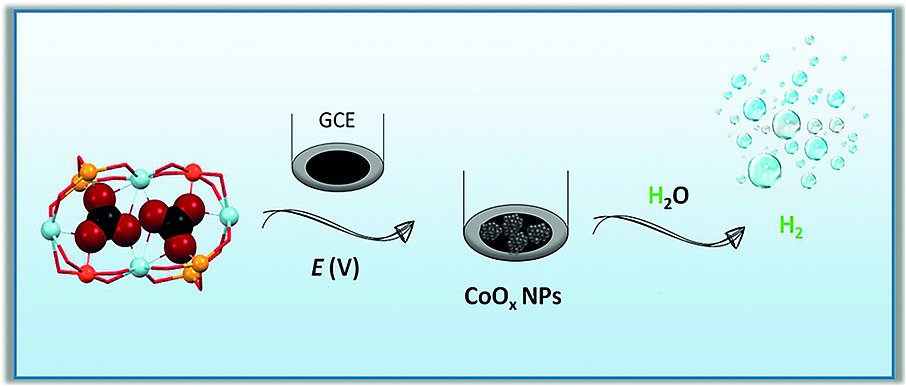
Electrochemical water reduction by employing first‐row transition‐metal nanoparticles (NPs) constitutes a sustainable way for the generation of H2. We have synthesized Co‐based NPs from a molecular CoII/CoIII precursor after its reductive decomposition at –1.86 V versus NHE in different organic solvents. These NPs are able to electrochemically reduce water at pH 14. SEM, EDX and XPS analyses have allowed the determination of the chemical nature of the as‐deposited NPs: CoO when using MeCN as the solvent and CoO(OH) when employing either dichloromethane (DCM) or MeOH. After 2 h of constant polarization at 10 mA cm–2, the electrocatalytic activity of the NPs obtained in MeCN and DCM decreases, whereas it increases for those obtained in MeOH. In this solvent, the overpotential is reduced by 215–220 mV and the specific current density is tripled. Interestingly, during this activation process in MeOH, the precursor CoO(OH) NPs are converted into Co(OH)2. The implications of these results in the context of the current research in the field are also discussed.