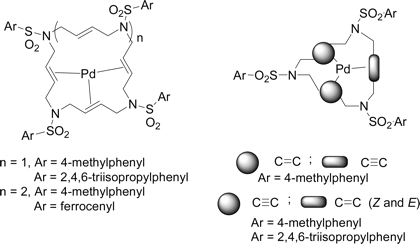
A novel type of chiral and stable palladium(0) complexes of polyunsaturated aza-macrocyclic ligands were prepared and fully characterized by means of NMR spectroscopy and X-ray diffraction. Fifteen-membered alkene-alkyne type ligands as well as 20- and 25-membered polyolefinic ligands showed a preference for tricoordination with the metal. The stereochemical complexity of these complexes is related to the different isomers that can be formed by complexation of the metal to either of the two faces of each of the olefins involved. The palladacyclopropane formulation of the palladium-olefin interaction offers a clear picture of the stereogenicity of the olefin carbon atoms when they are coordinated to the metal. The preorientation of the macrocyclic ligand and the prepositioning of the olefinic bonds for complex formation facilitated by six-membered chelate rings explain the stability of the structures. The conformation of the palladacyclohexanic rings is found to be crucial in the stereoisomers formed. These structural characteristics of the complexes have been studied in solution by NMR spectroscopy and in the solid state by X-ray diffraction analysis.