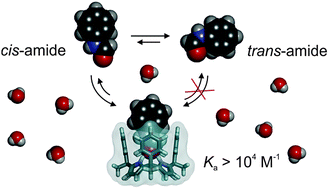
We describe the synthesis of a tetrapyridinium phenyl extended calix[4]pyrrole that is soluble in neutral water solution at mM concentrations. We show that, in pure water, the synthesized calix[4]pyrrole receptor selectively binds the cis-(E) conformers of secondary N-phenyl-amides and tertiary N-methyl-N-phenyl-formamide with binding affinities larger than 103 M−1. The conformational selectivity is remarkable owing to the energetic preference of amides to adopt the trans-(Z) conformation in solution. In this respect, we used two binding models for the mathematical analyses of the titration data and calculated apparent and intrinsic binding constants. The combined action of hydrogen bonding and the hydrophobic effect that operates in the binding of the amides in water is responsible for the large affinities displayed by the receptor.