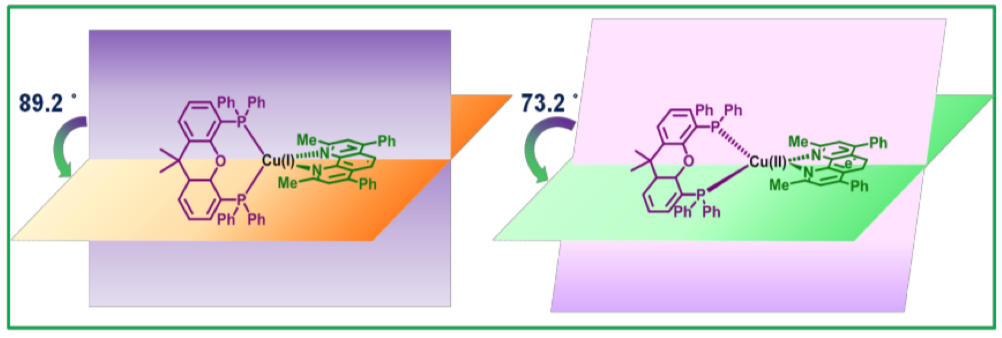
We report the light-induced electronic and geometric changes taking place within a heteroleptic Cu(I) photosensitizer, namely [(xant)Cu(Me2phenPh2)]PF6 (xant = xantphos, Me2phenPh2 = bathocuproine), by time-resolved X-ray absorption spectroscopy in the ps-μs time-regime. Time-resolved X-ray absorption near edge structure (XANES) and extended X-ray absorption fine structure (EXAFS) analysis enabled the elucidation of the electronic and structural configuration of the copper centre in the excited state as well as its decay dynamics in different solvent conditions with and without triethylamine acting as a sacrificial electron donor. A three- fold decrease in the decay lifetime of the excited state is observed in the presence of triethylamine showing the feasibility of the reductive quenching pathway in the latter case. A prominent pre-edge feature is observed in the XANES spectrum of the excited state upon metal to charge ligand transfer transition showing an increased hybridization of the 3d states with the ligand p orbitals in the tetrahedron around the Cu centre. EXAFS and Density Functional Theory illustrate a significant shortening of the Cu-N and an elongation of the Cu-P bonds together with a decrease in the torsional angle between the xantphos and bathocruproine ligand. This study provides mechanistic time-resolved understanding for the development of improved heteroleptic Cu(I) photosensitizers, which can be used for the light-driven production of hydrogen from water.