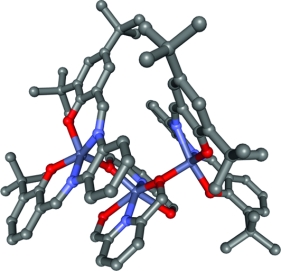
We report here a full study on the templating properties of a series of Zn(salphen) complexes toward pyridine-alcohol substrates by selective activation of the OH group providing trinuclear supramolecular assemblies as products. The templated process was investigated by various techniques including NMR spectroscopy, MALDI-TOF mass spectrometry and X-ray diffraction studies. A range of substrates were tested to reveal the scope and limitations of this process. The successful examples furnish remarkable trinuclear supramolecular assemblies comprising a non-symmetrical central unit made up of two monodeprotonated pyridine-alcohol ligands that coordinate a ZnII ion. The anionic oxygen atoms of this central Zn complex in turn are both associated with a Zn(salphen) unit. A number of model compounds were also prepared and substantiated the view that the first step in the templated mechanism involves the coordination of the alcohol function to the ZnII centre of the salphen complex. Hereafter, a double proton transfer to the salphen ligand occurs with simultaneous formation of the nonsymmetrical central complex, which combines with two Zn(salphen) modules through coordinative Zn-O bonds. The latter may be regarded as effective supramolecular protecting groups: stability studies performed in more polar media have revealed the full reversibility of the templated process. The reactivity of the Zn(salphen) complex towards alcoholic substrates may be useful in Lewis acid mediated synthesis.