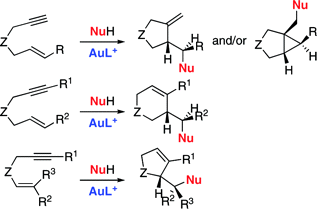
Gold(I)-catalyzed addition of carbon nucleophiles to 1,6-enynes gives two different type of products by reaction at the cyclopropane or at the carbene carbons of the intermediate cyclopropyl gold carbenes. The 5-exo-dig cyclization is followed by most 1,6-enynes, although those bearing internal alkynes and alkenes react by the 6-endo-dig pathway. The cyclopropane versus carbene site-selectivity can be controlled in some cases by the ligand on the gold catalyst. In addition to electron-rich arenes and heteroarenes, allylsilanes and 1,3-dicarbonyl compounds can be used as the nucleophiles. In the reaction of 1,5-enynes with carbon nucleophiles, the 5-endo-dig pathway is preferred.