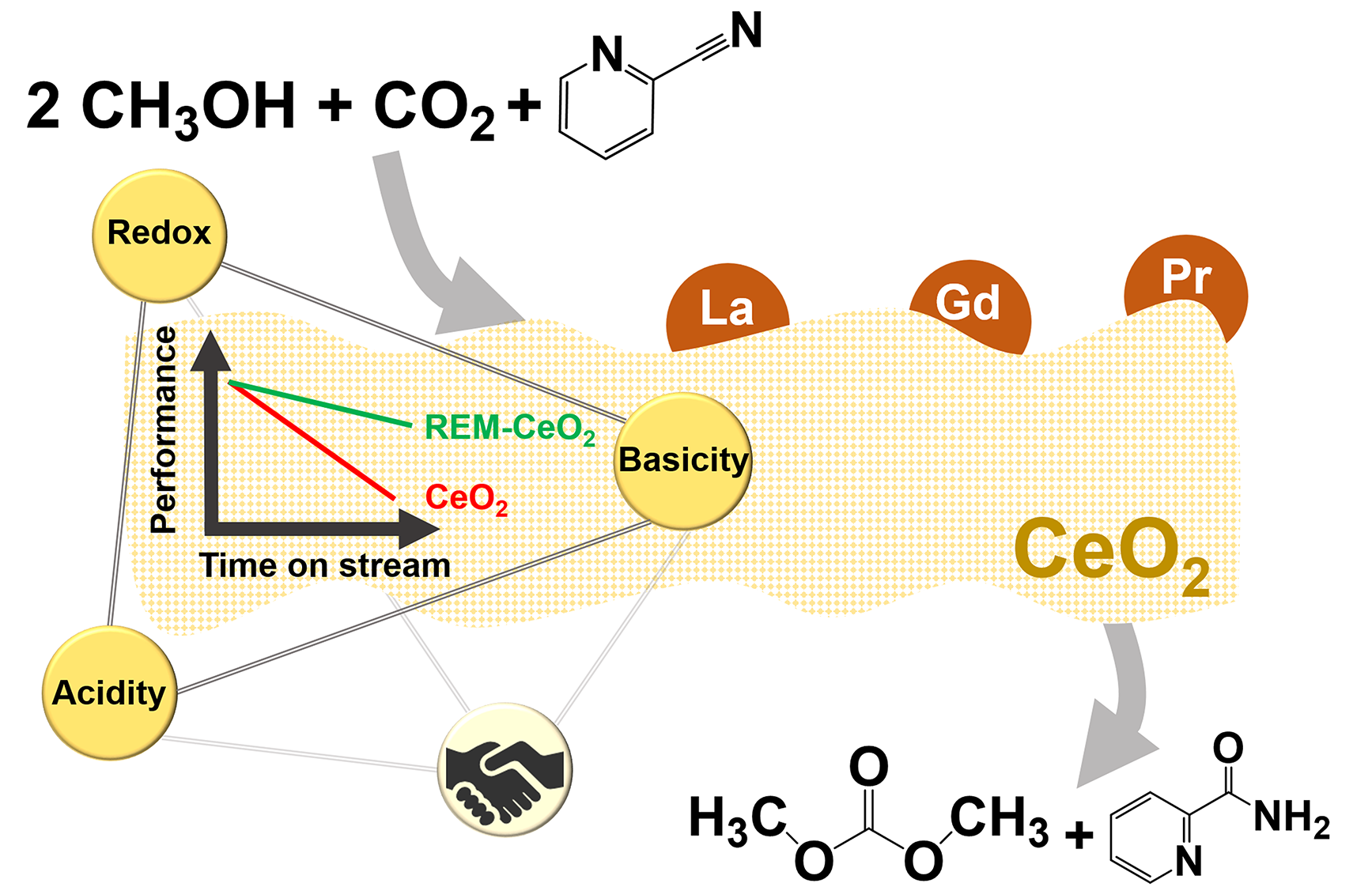
The unmatched efficiency of the direct dimethyl carbonate (DMC) synthesis from CO2 and methanol over CeO2 catalysts in the presence of an organic dehydrating agent (2-cyanopyridine, 2-CP) was recently reported with high DMC yield (>90%) in both batch and continuous operations. However, the CeO2 catalyst gradually deactivates in the time-scale of days due to suggested surface poisoning by 2-picolinamide (2-PA) produced by hydration of 2-CP. This work seeks for active and stable CeO2-based catalysts and aims to understand the material factors influencing the catalytic performance. Surface modification of CeO2 by the addition of rare earth metal (REM) was found effective to improve the catalyst stability. Surface basicity and reducibility of the Ce4+species play important roles in preventing catalyst deactivation by stabilizing the reactive methoxy species in comparison to the poisoning species (2-PA or species alike). This has been evidenced by in situ ATR-IR spectroscopy. CeO2 materials promoted with 1 wt% rare earth metals (La, Gd, and Pr) greatly enhanced the catalyst stability while retaining the high catalytic activity of CeO2. Among them, 1 wt% Pr promotion to CeO2 was the most effective, affording 35% higher DMC yield in comparison to bare CeO2 after 150 h time on stream under the optimized reaction condition of 30 bar and 120 °C.