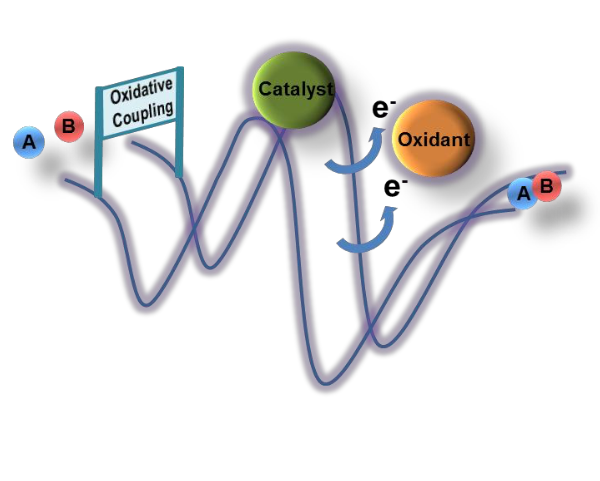
Oxidative coupling reactions, where two electrons are released from the reactants and trapped by an oxidant, have arisen as a versatile alternative to cross-coupling in chemical synthesis. Despite the large number of experimental reports on the process, a clear mechanistic picture is only starting to emerge. In this perspective, we highlight the contribution from DFT calculations to the computational characterization of this mechanism. Oxidative coupling processes have been reported differing in both the catalyst (radicals, precious metals or earth-abundant metals) and the oxidant. We have found more useful to classify them according to the oxidant used, as metal-based oxidants and metal-free oxidants seem to favor different mechanistic variations. All steps in the full catalytic cycle are analyzed, and issues concerning selectivity and influence of the oxidant are considered.