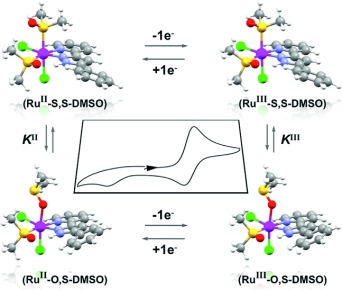
The structures, spectroscopic properties, electrochemistry, and reactivity of two new isomeric [Ru(Cl)2(L)(DMSO)2] complexes are reported [L is the nonsymmetric chelating ligand 5-phenyl-3-(2-pyridyl)-1H-pyrazole (H3p), DMSO = dimethyl sulfoxide]. It is shown that there is S-to-O linkage isomerization of the ruthenium(II) sulfoxide complexes upon one electron oxidation. The thermodynamic and kinetic properties of this linkage isomerization differ for each isomeric complex and depend strongly on the protonation grade of the ligand backbone. The corresponding data for these processes were evaluated from electrochemical data. A photolytic reaction of either of these isomers in chloroform is presented and leads to the substitution of one DMSO ligand and the formation of a third complex, [Ru(Cl)3(L)(DMSO)]. The remaining coordinated DMSO ligand retains its binding mode through the sulfur atom to a ruthenium(III) metal center.