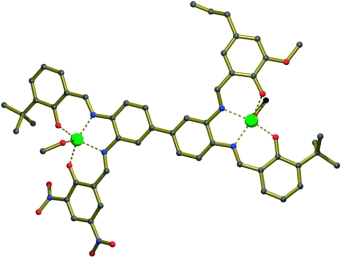
A metal-templated, stepwise approach toward fully nonsymmetrical bis-metallosalphen complexes is described. The synthesis comprises the selective monometalation of diimine precursors and subsequent introduction of the second metal ion by using various salicylaldehyde reagents. In the case of a bis-Zn(salen) derivative, the presence of different peripheral substituents on the two metallosalen units gave rise to a large difference in kinetic stability, which was used to selectively demetalate the more labile site. This result shows promise for the preparation of heterobimetallic structures. The first X-ray molecular structure of a completely nonsymmetrical bis-salphen complex is also reported.