Our research will center on various aspects of the catalytic conversion of ethylene into a given α-olefin, as exemplified by the selective production of 1-hexene. If compared to the classical oligomerization, this class of reactions may lead to a more efficient hydrocarbon synthesis, in part by obviating the need for the fractional separation. Our efforts will also focus on understanding the behavior of the metallacyclic intermediates involved in the unique mechanism of this transformation.
Olefin polymerization and oligomerization
Olefin polymerization and oligomerization has traditionally been accomplished using transition metal catalysts (or initiators) operating through a sequential coordination-insertion of olefins into a growing polymer chain. With the α-H elimination as a chain-termination step, the process can be used for the production of the valuable low MW a-olefins, as seen, for example, in the Shell Higher Olefin Process. However, the statistical nature of the process leads to a distribution of chain lengths; a separation step is thus required to obtain a specific α-olefin. This has led to a search for catalysts capable of selectively transforming ethylene (or other olefins) into a specific higher α-olefin.
Efficient chromium catalysts for the trimerization of ethylene to form 1-hexene have been developed, with the first industrial plant based on this method brought on-line in 2004. The catalysts in such a process operate through a metallacycle intermediate, with the selectivity attributed to the preferential olefin elimination from a 7-membered metallacycle.
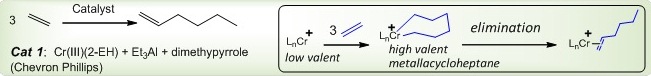
Despite significant advances made by several groups in unraveling the mechanism of olefin trimerization, certain aspects remain poorly understood. We will use the related diamagnetic Ti models (also active in trimerization) to observe and characterize both the low-valent electrophilic form of the catalyst, and to trap the resulting metallacyclic species; selective elimination of the olefin from a 7-membered cycle will be studied, with the mechanistic knowledge then applied to the design of new catalysts.
Recently, certain types of Cr catalysts were found to carry out ethylene tetramerization to give 1-octene. Although the overall mechanism appears to be similar to that for the trimerization, the exact reasons for releasing 1-octene rather than 1-hexene remain uncertain and the process appears to be highly ligand-dependent. One possibility is the relative stabilization of the 7-membered ring at the expense of the 9-membered metallacycle; elimination from this latter would then give 1-octene (path A). An intriguing bimetallic mechanism (path B) has also been proposed to account for such selectivity.

To probe the latter proposal, our group will synthesize a series of bimetallic metal complexes; their behavior in ethylene trimerization/tetramerization will then be tested. In addition to a search for a truly efficient and selective formation of 1-octene, a larger question stands as to whether the concept could be extended to selectively produce an α-olefin chain more than 8-carbon long.