Our research aim is the rational improvement and design of heterogeneous catalytic processes based on solid comprehension of the elementary reaction steps and the transformation mechanisms in order to minimize risks and damages of chemical processes on environment while achieving high efficiencies in conversion, selectivity, and energy usage. This ambitious goal is pursued by an interdisciplinary experimental-theoretical approach; in situ spectroscopy and theoretical modeling for solid understanding of the processes as well as reaction engineering to maximize the efficiencies.
Powerful in situ spectroscopic tools for studying solid-fluid (gas, liquid, and supercritical fluid) interfaces are being developed and applied to gain key insights into the reaction mechanisms with the aid of static and dynamic density functional theory (DFT) calculations for the interpretation of experimental results.
Among others, one of the major reactions we currently focus on is heterogeneous catalytic conversion of CO2 into fuels and useful chemicals at high efficiency, so that the carbon-cycle, which has generally an open end due to burning of fossil fuels and release as CO2 into atmosphere, can be closed for sustainable development.
In situ/operando Spectroscopy
Identification of involved species during catalytic reactions and the reaction mechanisms under practically relevant in situ/operando conditions (often under pressure at high temperature) is the crucial step for a rational development of catalytic processes. Heterogeneous catalysis occurs at solid-fluid (gas, liquid, and supercritical fluid) interfaces and the sensitive and selective detection of chemical processes at the surfaces is required. We develop spectroscopic tools for such demanding investigations and apply them to rationally improve catalysts for our target reactions.
Vibrational (IR and Raman) spectroscopy is one of our main spectroscopic tools. We design unique cells so that we can study catalytic reactions in a highly time-resolved and sometimes in a space-resolved manner to gain new insights into the mechanisms, kinetics, and chemical gradients of catalytic reactions. Various detection techniques such as diffuse reflectance infrared Fourier transform spectroscopy (DRIFTS, Figure 1), polarization-modulation infrared-reflection absorption spectroscopy (PM-IRRAS, Figure 2), attenuated total reflection infrared spectroscopy (ATR-IRS), and Raman spectroscopy, are utilized and combined to gain insights into the chemical processes occurring on the surface and in the bulk of catalysts as well as in the fluids. The obtained information is further enriched and complemented by that gained by structure and electronic-state sensitive methods such as in situ X-ray diffraction (XRD) and absorption fine structure (XAFS) measured at synchrotrons.
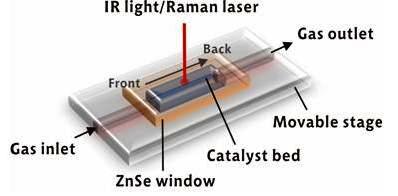
Figure 1. Probing chemical gradients along the axial direction of a catalyst bed by space- and time-resolved DRIFT-Raman spectroscopy
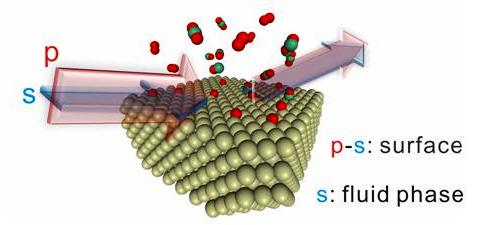
Figure 2. Time-resolved, simultaneous but separate detection of chemical species on a catalyst surface and in the fluid above the surface by in situ PM-IRRAS
In situ/operando spectroscopic measurements, however, yield often very complex spectral features and hinder the selective monitoring of catalytic active species and sites due to high concentrations of catalytically non-active (spectator) or non-relevant species such as solvent. The complication is further pronounced by the highly dynamic nature of chemical systems at high temperature and pressure.
To overcome these difficulties, we apply a powerful technique, modulation excitation spectroscopy (MES), and combining it with various spectroscopic tools to greatly enhance their ‘sensitivity’ (thus the time-resolution) and introduce ‘selectivity’ into the tools to monitor only what we would like to observe (e.g. catalytic active sites). MES utilizes periodic perturbation of a parameter, such as concentration and temperature, to influence the concentration of the species of the interests and the kinetics of the chemical interactions and reactions. After a mathematical treatment, we can monitor only the key species and their kinetics sensitively and selectively. We apply MES to study complex reaction networks and also extend the scope of MES by further developing the underlying theory to study complex chemical problems.
Reaction Engineering and Microreaction Systems
Reaction engineering plays a key role in maximizing of the potential of catalysts by optimizing reaction parameters such as a reactor type and residence time. These parameters need to be carefully taken care when the performance of catalytic reactions is evaluated. Also, microreactors offer unique opportunity in chemical analysis and process intensification. When appropriate materials are used, they allow direct analysis on the reactor of all the involved chemical species and their gradients. Particularly for high-pressure reactions, the safety issues are greatly improved using such reactors. Currently, we develop microreaction systems for high pressure and high temperature applications.
Theoretical Modeling
Molecular modeling is a very rapidly growing field thanks to the ever-increasing computational power. In combination with efficient theoretical accounts such as DFT with and without the scheme of QM/MM, large systems including catalytic interfaces and solvents can be simulated. The accuracy given by such methods has made them often to be the ‘must’ for interpreting the spectroscopic data. Also powerful methodologies such as metadynamics can be utilized to study reaction pathways in a computationally efficient manner by accessing the underlying free energy surface of the chemical processes (Figure 3), clarifying the reaction mechanisms and identify a key reaction steps such as the rate-limiting step.
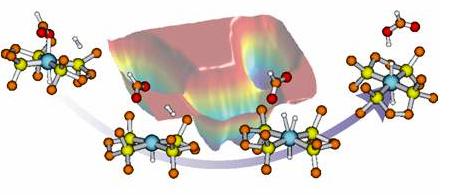
Figure 3. Elucidation of reaction pathways and mechanisms of Ru-complex catalyzed
CO2 hydrogenation by spectroscopy and ab initio metadynamics