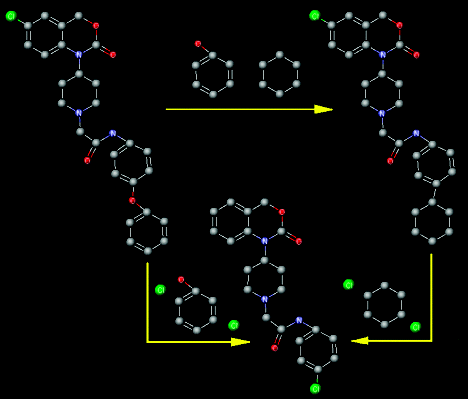
In this paper, we present the advantages of using data fusion of similarity and dissimilarity measurements for the development of quantitative structure-activity relationship models. Nonisomorphic fragments extracted in the matching process were considered to obtain dissimilarity values employed for correcting similarity measurements, thus, leading to finer chemical information. The purpose was to correlate similarity and dissimilarity matrices with pharmacological activities of drugs (the inhibitory capacity presented by 30 benzoxazinone derivatives for the NPY Y5 receptor). Wiener and hyper-Wiener descriptors computed over distance and weighted distance matrices were used for the calculation of dissimilarity values. A comparison with classical and fingerprints-based similarity was also carried out. The best approaches were achieved by means of dissimilarity and of fusion data spaces that take into account isomorphic and nonisomorphic information (Q2 = 0.88, SECV = 0.18, slope = 1.06, and intercept = 0.09). The study of anomalous behavior presented by some compounds was also undertaken.