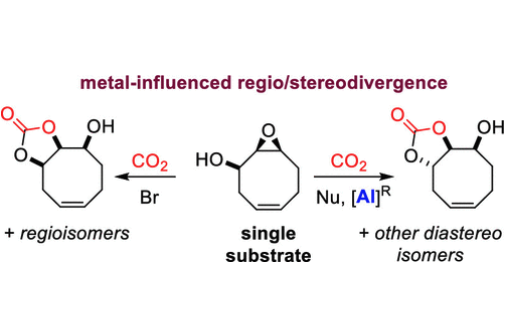
Valorization of carbon dioxide into organic molecules using catalytic approaches has witnessed an upsurge in recent years. Here, the influence of an Al(III) aminotriphenolate complex on the regio- and stereochemical features of the coupling between carbon dioxide and a cyclic epoxy alcohol has been studied. Three distinct bicyclic carbonate products were produced from a single starting material, depending on the catalytic conditions. The proposed carbonate configurations were examined by solution and solid-phase techniques, including NMR spectroscopic and X-ray crystallographic analyses. Control experiments combined with DFT calculations provide a rationale for the distinct catalytic manifolds observed in the presence and absence of the Al(III) complex.