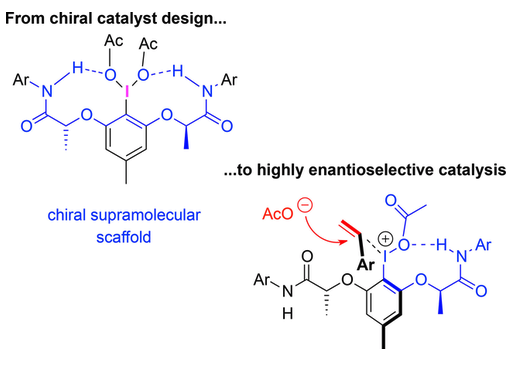
Chiral aryliodine(III) reagents have provided an advanced concept for enantioselective synthesis and catalysis. With the advent of chiral iodine(I/III) catalysis, many different structures have been explored in the area. The currently most prominent catalyst design is based on a resorcinol core and the attachment of two lactic side chains bearing ester or amide groups. This enables a privileged modular catalyst synthesis, in which fine‐tuning with respect to the specific reaction requirement is straightforward. The present overview summarizes the structural variation and optimization of such chiral aryliodine catalysts and discusses structural properties of the active iodine(III) catalyst states. The status quo of enantioselective iodine(I/III) oxidation catalysis with respect to intramolecular and intermolecular reaction control is reviewed, and specific aspects of the individual catalytic cycles are discussed.