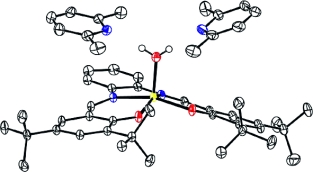
The binding properties of various Zn(salphen) complexes with a range of substituted pyridine ligands has been studied in detail using NMR and UV/Vis spectroscopy, X-ray diffraction analysis and molecular modelling studies. The combined results clearly demonstrate that subtle differences in the ortho-substitution pattern on the pyridine ring have a dramatic effect on the strength of the Zn-Npyr interaction. In the case of a double ortho-substitution, the pyridine donor is unable to bind to the Zn(salphen) complex as a result of the rigid geometry around the Zn ion and the steric repulsion that results upon ligation. The steric requirements of the pyridine ligand infers a preferred positioning of its pendant groups as to minimize repulsive interactions with the salphen ligand. The crystallization of two separate Zn(salphen) complexes in the presence of 2,6-dimethylpyridine has furnished two remarkable 4-component supramolecular structures in which the Zn ion is associated with a water ligand that in turn is surrounded by two pyridine H-bond acceptors; a result that relates to the inability of this pyridine ligand to directly interact with the Zn metal centre.