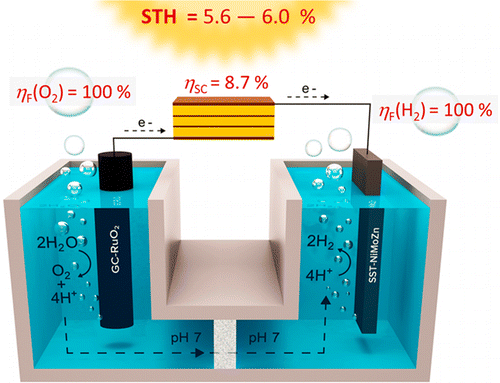
We report a photovoltaics-electrochemical (PV-EC) assembly based on a compact and easily processable triple homojunction polymer cell with high fill factor (76%), optimized conversion efficiencies up to 8.7%, and enough potential for the energetically demanding water splitting reaction (Voc = 2.1 V). A platinum-free cathode made of abundant materials is coupled to a ruthenium oxide on glassy carbon anode (GC-RuO2) to perform the reaction at optimum potential (ΔE = 1.70–1.78 V, overpotential = 470–550 mV). The GC-RuO2 anode contains a single monolayer of catalyst corresponding to a superficial concentration (Γ) of 0.15 nmol cm—2 and is highly active at pH 7. The PV-EC cell achieves solar to hydrogen conversion efficiencies (STH) ranging from 5.6 to 6.0%. As a result of the solar cell’s high fill factor, the optimal photovoltaic response is found at 1.70 V, the minimum potential at which the electrodes used perform the water splitting reaction. This allows generating hydrogen at efficiencies that would be very similar (96%) to those obtained as if the system were to be operating at 1.23 V, the thermodynamic potential threshold for the water splitting reaction.