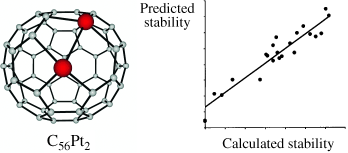
A systematic search of the regioisomers of the heterofullerenes, C57Pt2 and C56Pt2, has been carried out by means of density functional calculations to find the most stable structures. Both heterofullerenes incorporate two metal atoms into the fullerene surface. In the case of C57Pt2, one platinum atom substitutes one carbon atom of C60 and the other platinum atom replaces a C-C bond, whereas in C56Pt2 each platinum atom replaces one C-C bond. Several geometric factors were studied, three of which have particularly important effects on the relative stabilities of the regioisomers: the Pt-Pt separation, the number of C-C bonds remaining after substitution, and the type of C-C bond that is substituted. All these factors indicate that the deformation of the carbon framework is a general factor that governs the relative stabilities of the regioisomers. Because a high number of factors affect the stability of the heterofullerenes we also used chemometric techniques in this study. Partial least-squares (PLS) regression was used to establish the structure-energy relationships of C57Pt2 and C56Pt2 heterofullerenes. The understanding gained of the factors that affect the relative isomers stabilities has allowed us to predict the stabilities of larger disubstituted carbon cages, for example, C81Pt2 heterofullerene.