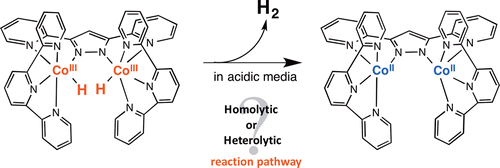
A dinuclear Co complex with bis(pyridyl)pyrazolato (bpp–) and terpyridine (trpy) ligands, [CoIII2(trpy)2(μ-bpp)(OH)(OH2)]4+ (14+), undergoes three-electron reduction by cobaltocene in acetonitrile to produce 1+, which is in the protonation equilibrium with the CoIICoIII–hydride complex, and the further protonation of the hydride by trifluoroacetic acid yields hydrogen quantitatively. The kinetic study together with the detection of the CoIICoIII-hydride complex revealed the mechanism of the hydrogen production by the reaction of 1+ with trifluoroacetic acid.