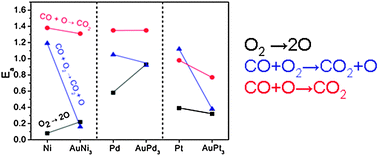
Gold alloys have been reported to be active in very low temperature oxidation of CO. The reasons for the better performance of AuNi with respect to Ni(111) surfaces were reported recently by indicating an active role of gold. Our results show that for this alloy, the CO oxidation molecular channel is responsible for the low temperature performance but the remaining oxygen on the surface is tightly bound and the process is not catalytic at low to room temperatures. The contribution from the associative path is also improved for the rest of the series: AuPd and AuPt. Only in the latter case, an important reduction of the light-off temperature of the catalytic path is observed.