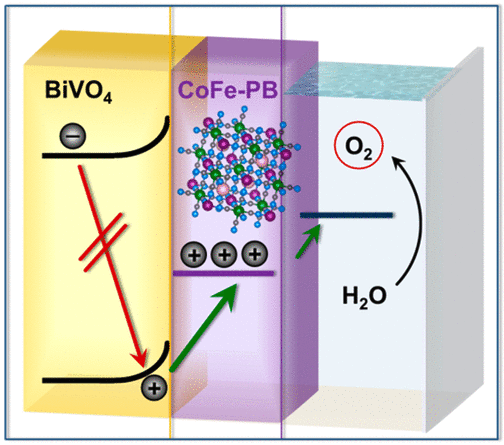
Catalyst modification of metal oxide photoanodes can result in markedly improved water oxidation efficiency. However, the reasons for improvement are often subtle and controversial. Upon depositing a CoFe Prussian blue (CoFe-PB) water oxidation catalyst on BiVO4, a large photocurrent increase and onset potential shift (up to 0.8 V) are observed, resulting in a substantially more efficient system with high stability. To elucidate the origin of this enhancement, we used time-resolved spectroscopies to compare the dynamics of photogenerated holes in modified and unmodified BiVO4 films. Even in the absence of strong positive bias, a fast (pre-ms), largely irreversible hole transfer from BiVO4 to CoFe-PB is observed. This process retards recombination, enabling holes to accumulate in the catalyst. Holes in CoFe-PB remain reactive, oxidizing water at a similar rate to holes in pristine BiVO4. CoFe-PB therefore enhances performance by presenting a favorable interface for efficient hole transfer, combined with the catalytic function necessary to drive water oxidation.