In this communication, I will showcase some of the research I conducted after leaving ICIQ, switching from the simulation of nonbiological catalysts to biological ones (i.e., enzymes).
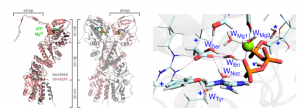
I will mainly focus on the work I carried out during my current position in Pavia, whereby I was able to model and justify the peculiar reactivity of the (homodimeric) mitochondrial chaperone Trap1 (Figure 1; right). This molecular machine—whose activity and/or expression levels are notably altered in several cancers[1]—oversees the correct folding of bound “client” proteins, a feat which it achieves through extensive conformational rearrangement.[2] Kickstarting this rearrangement are two strictly sequential ATP hydrolyses, which can only occur once the chaperone adopts a distinctive asymmetric “closed” state (Figure 1; left):[2] intriguingly, the ATP molecule bound to the buckled protomer Buc is cleaved first (and, even so, sluggishly); conversely, the straight protomer Str remains catalytically inert until the first hydrolysis has triggered its own buckling.
Using a previously tested recipe,[3] classical molecular dynamics (MD) proved that, in Str, nucleophilic water WatNuc is more frequently sequestered, via tighter hydrogen bonds, by a (conserved) tyrosine and its vicinal water WatTyr: “reactive poses” (Figure 1; left) are thus more frequent in Buc. Hybrid semiempirical quantum-classical (QM/MM) MD with umbrella sampling, benchmarked with density functional theory, thence confirmed that looser WatNuc sequestration is enough to lower the free energy required for hydrolysis.
If time allows, I will conclude with a summary of our current work on SARS-CoV-2 proteins.
Figure 1. (Left) Overview of zebrafish Trap1 (zTrap) with its protomers in the asymmetric “closed” state. (Right) One of zTrap’s active sites just prior to ATP hydrolysis (green sphere: Mg2+).
[1] Sánchez-Martín, C.; Serapian, S. A.; Colombo, G.; Rasola, A., Front. Oncol. 2020, 10, 1177.
[2] Elnatan, D.; Betegon, M.; Liu, Y.; Ramelot, T.; Kennedy, M. A.; Agard, D. A., eLife 2017, 6, e25235.
[3] Serapian, S. A.; van der Kamp, M. W., ACS Catal. 2019, 9, 2381.
This event will be recorded and uploaded on ICIQ’s YouTube Channel.
If you would like to attend this talk, please, register here!