Prof. Antoni Llobet (Institute of Chemical Research of Catalonia, ICIQ), along with an International team of scientists, designs a ruthenium catalyst for the most effective oxidation of water to date.
For the first time, a research group led by Antoni Llobet (Institute of Chemical Research of Catalonia, ICIQ, Spain), Timofei Privalov (Stockholm University, Sweden) and Licheng Sun (Dalian University of Technology, China) synthesises a compound able to catalyse the oxidation of water and produce oxygen with an efficiency comparable to that of the Photosystem II, the natural catalyst of green plants.
The oxidation of water is the first reaction that occurs in water-splitting and it is the most important and complex step of the whole process.
Water-splitting:
1. Water Oxidation: 2 H2O → 4H+ + 4 e–+ O2
2. Hydrogen production: 4H+ + 4 e–→ 2 H2
In nature, this process is catalysed by a cluster of manganese and calcium (Mn4CaO5) located in the Photosystem II (PSII) of green plants. The bio-inspired synthesis of a stable compound able to mimic the activity of PSII effectively has been a great challenge until now.
Now, Antoni Llobet and his research team publish in Nature Chemistry* the synthesis and mechanism of the first compound capable of oxidizing water with an efficiency comparable to that of the natural catalyst.
Up until now, the highest turnover frequency achieved by synthetic catalysts (TOF) did not exceed 5 s-1, far below the 100-400 s-1 needed to be comparable to the natural catalyst. The ruthenium compound designed by Llobet’s team reaches 300 s-1 leading to the formation of 0.72 litres of oxygen per milligram of catalyst.
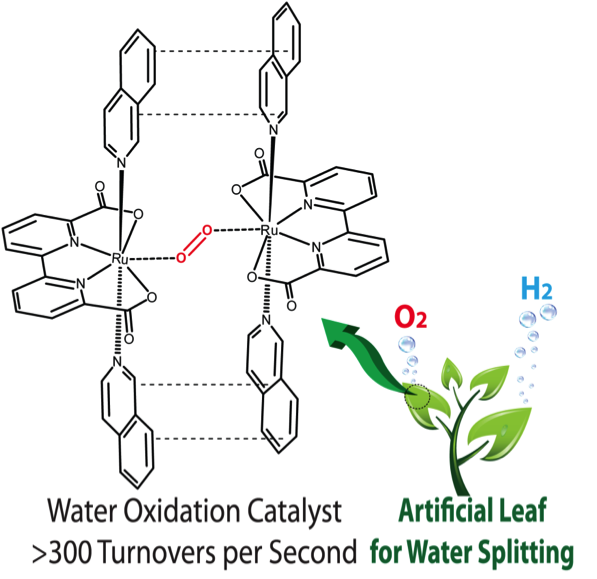
This research is an important stepping-stone in the search for a viable process of hydrogen production from water. Water oxidation provides, besides oxygen, the protons and electrons required in the subsequent generation of hydrogen.
Hydrogen production is one of the fields of research that generates more interest within the scientific community, since hydrogen could be a sustainable alternative to fossil fuels, which are responsible for the greenhouse effect.