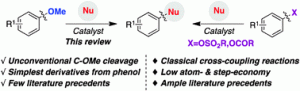
A paper published by Prof. R. Martin et al. in Chemical Society Reviews has been the most read article for the last 30 days.
The review highlights the prospective impact of simple ethers as aryl halide surrogates in the cross-coupling arena via C-O bond-activation.
Metal-catalyzed activatoin of ethers via C-O bond cleavage: a new strategy for molecular diversity
J. Cornella, C. Zarate, R. Martin
Chem. Soc. Rev., 2014 (Advance Article)
Another paper by Prof. R. Martin research group published in Journal of the American Chemical Society is in the list of most read articles in August.
The paper describes a Ni-catalyzed carboxylation of unactivated primary alkyl bromides and sulfonates with CO2 at atmospheric pressure. The method is characterized by its mild conditions and remarkably wide scope without the need for air- or moisture-sensitive reagents, which make it a user-friendly and operationally simple protocol en route to carboxylic acids.
Ni-Catalyzed Carboxylation of Unactivated Primary Alkyl Bromides and Sulfonates with CO2
Y. Liu, J. Cornella, R. Martin
J. Am. Chem. Soc., 2014, 136, 11212-11215