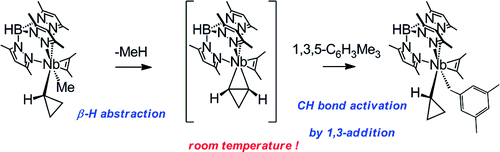
The methyl cyclopropyl hydrotris(3,5-dimethylpyrazolyl)borate complex TpMe2NbMe(c-C3H5)(MeCCMe) reacts smoothly with different alkylaromatics XH at 308 K to yield methane and TpMe2NbX(c-C3H5)(MeCCMe). NMR data show that for mesitylene and 1,4-dimethylbenzene, selective benzylic CH bond activation is observed, giving the benzyl cyclopropyl complexes TpMe2Nb(CH2Ar′)(c-C3H5)(MeCCMe) (Ar′ = 3,5-Me2C6H3, 4-MeC6H4, respectively). Selective arene CH bond activation is observed with 1,2-dimethylbenzene, yielding TpMe2Nb(3,4-Me2C6H3)(c-C3H5)(MeCCMe). With 1,3-dimethylbenzene, a 3:1 mixture of arene and benzylic CH activated products, TpMe2Nb(3,5-Me2C6H3)(c-C3H5)(MeCCMe) and TpMe2Nb(CH2-3-MeC6H4)(c-C3H5)(MeCCMe), is observed, translating to a 18:1 preference for aromatic versus benzylic CH bond activation on a per CH bond basis. Kinetic studies are consistent with rate-limiting intramolecular β-H abstraction of methane to yield a transient unsaturated η2-cyclopropene intermediate. This intermediate reacts rapidly with the aromatic or benzylic CH bond of the arene via 1,3-addition across a Nb(η2-cyclopropene) bond. DFT calculations suggest that the observed selectivities are a result of the steric influence of the methyl groups on the arene ring, which blocks activation of an ortho C-H bond.