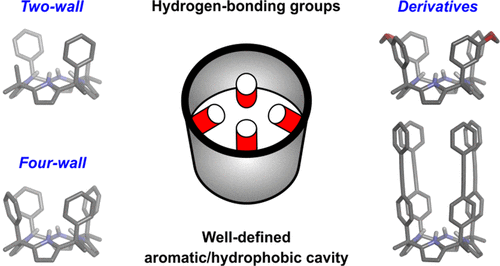
Proteins exhibit high-binding affinity and selectivity, as well as remarkable catalytic performance. Their binding pockets are hydrophobic but also contain polar and charged groups to contribute to the binding of polar organic molecules in aqueous solution. In the past decades, the synthesis of biomimetic receptors featuring sizable aromatic cavities equipped with converging polar groups has received considerable attention. “Temple” cages, naphthotubes, and aryl-extended calix[4]pyrroles are privileged examples of synthetic scaffolds displaying functionalized hydrophobic cavities capable of binding polar substrates. In particular, calix[4]pyrroles are macrocycles containing four pyrrole rings connected through their pyrrolic 2- and 5-positions by tetra-substituted sp3 carbon atoms (meso-substituents). In 1996, Sessler introduced the meso-octamethyl calix[4]pyrrole as an outstanding receptor for anion binding. Independently, Sessler and Floriani also showed that the introduction of aryl substituents in the meso-positions produced aryl-extended calix[4]pyrroles as a mixture of configurational isomers. In addition, aryl-extended calix[4]pyrroles bearing two and four meso-aryl substituents (walls) were reported. The cone conformation of “two-wall” αα-aryl-extended calix[4]pyrroles features an aromatic cleft with a polar binding site defined by four converging pyrrole NHs. On the other hand, “four-wall” αααα-calix[4]pyrrole isomers possess a deep polar aromatic cavity closed at one end by the converging pyrrole NHs. Because of their functionalized interior, aryl-extended calix[4]pyrroles are capable of binding anions, ion-pairs, and electron-rich neutral molecules in organic solvents. However, in water, they are restricted to the inclusion of neutral polar guests.
Since the early 2000s, our research group has been involved in the design and synthesis of “two-wall” and “four-wall” aryl-extended calix[4]pyrroles and their derivatives, such as aryl-extended calix[4]pyrrole cavitands and super aryl-extended calix[4]pyrroles. In this Account, we mainly summarize our own results on the binding of charged and neutral polar guests with these macrocyclic receptors in organic solvents and in water. We also describe the applications of calix[4]pyrrole derivatives in the sensing of creatinine, the facilitated transmembrane transport of anions and amino acids, and the monofunctionalization of bis-isonitriles. Moreover, we explain the use of calix[4]pyrrole receptors as model systems for the quantification of anion−π interactions and the hydrophobic effect. Finally, we discuss the self-assembly of dimeric capsules and unimolecular metallo-cages based on calix[4]pyrrole scaffolds. We comment on their binding properties, as well as on those of bis-calix[4]pyrroles having a fully covalent structure.
In molecular recognition, aryl-extended calix[4]pyrroles and their derivatives are considered valuable receptors owing to their ability to interact with a wide variety of electron-rich, neutral, and charged guests. Calix[4]pyrrole scaffolds have also been applied in the development of molecular sensors, ionophores, transmembrane carriers, supramolecular protecting groups and molecular containers modulating chemical reactivity, among others. We believe that the design of new calix[4]pyrrole receptors and the investigation of their binding properties may lead to promising applications in many research areas, such as supramolecular catalysis, chemical biology and materials science. We hope that this Account will serve to spread the knowledge of the supramolecular chemistry of calix[4]pyrroles among supramolecular and nonsupramolecular chemists alike.