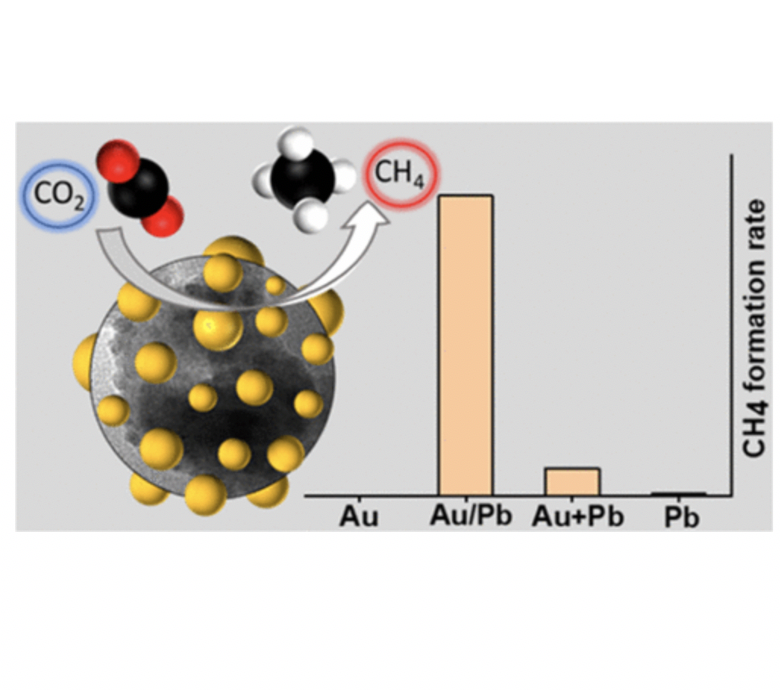
The electrochemical conversion of carbon dioxide (CO2) to high-value chemicals is an attractive approach to create an artificial carbon cycle. Tuning the activity and product selectivity while maintaining long-term stability, however, remains a significant challenge. Here, we study a series of Au–Pb bimetallic electrocatalysts with different Au/Pb interfaces, generating carbon monoxide (CO), formic acid (HCOOH), and methane (CH4) as CO2 reduction products. The formation of CH4 is significant because it has only been observed on very few Cu-free electrodes. The maximum CH4 formation rate of 0.33 mA cm–2 was achieved when the most Au/Pb interfaces were present. In situ Raman spectroelectrochemical studies confirmed the stability of the Pb native substoichiometric oxide under the reduction conditions on the Au–Pb catalyst, which seems to be a major contributor to CH4 formation. Density functional theory simulations showed that without Au, the reaction would get stuck on the COOH intermediate, and without O, the reaction would not evolve further than the CHOH intermediate. In addition, they confirmed that the Au/Pb bimetallic interface (together with the subsurface oxygen in the model) possesses a moderate binding strength for the key intermediates, which is indeed necessary for the CH4 pathway. Overall, this study demonstrates how bimetallic nanoparticles can be employed to overcome scaling relations in the CO2 reduction reaction.