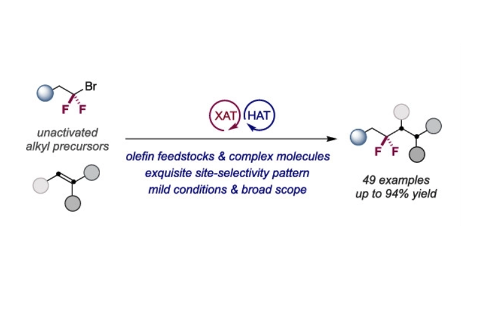
Herein, we report a modular catalytic technique that streamlines the preparation of gem-difluoroalkanes from unactivated sp3 precursors. The method is characterized by its simplicity, generality, and site selectivity, including the functionalization of advanced intermediates and olefin feedstocks. Our approach is enabled by a cooperative interplay of halogen- and hydrogen-atom transfer, thus offering a new entry point to difluorinated alkyl bioisosteres of interest in drug discovery.