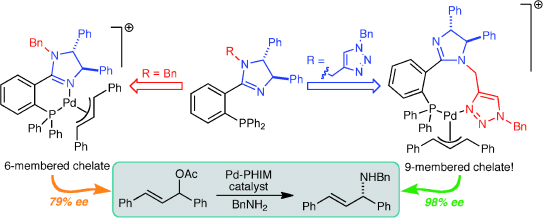
Phosphinoimidazoline (PHIM) ligands bearing a triazolylmethyl substituent at the sp3 nitrogen atom in the imidazoline ring lead to highly improved enantioselectivity (up to 99% ee) in allylic substitution reactions with respect to analogous ligands with substituents lacking the triazole unit. NMR and theoretical studies support a shift in the coordination mode of the PHIM ligand to palladium, triggered by a very favourable interaction with the triazole unit.