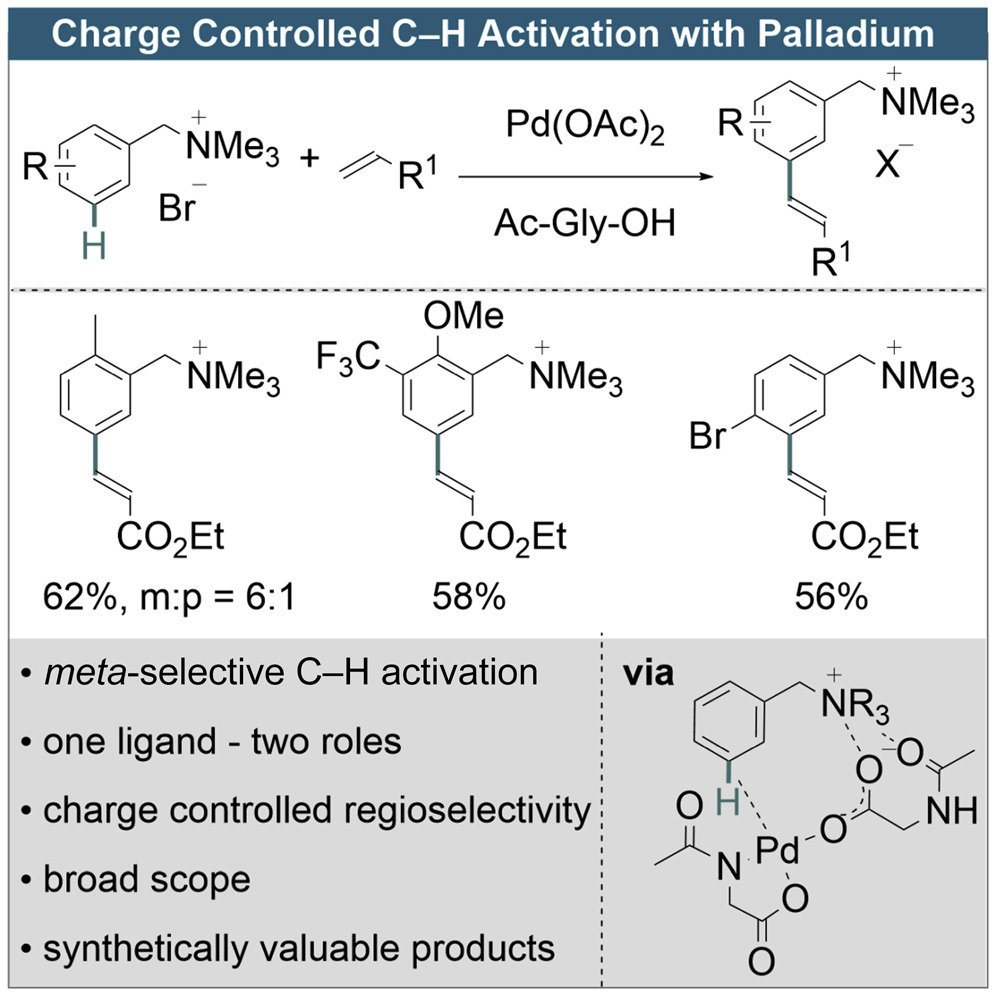
The regioselective C–H activation of arenes remains one of the most promising techniques to access highly important functionalized motifs. Such functionalizations can generally be achieved through directed and non-directed processes. The directed approach requires a covalently attached directing group (DG) on the substrate to induce reactivity and selectivity, and therefore intrinsically leaves a functional group at the point of attachment within the molecule, even after the tailored DG has been removed. Conversely, non-directed methods typically suffer from regioselectivity issues, especially for unbiased substrates. Herein, we report a unique approach to address these challenges in palladium catalysis that employs weak charge-charge and charge-dipole interactions to enable the meta-selective activation/olefination of arenes. The charged moiety could easily be converted to uncharged simple arenes by hydrogenation or cross-coupling. In-depth mechanistic studies prove that the charge is responsible for the observed selectivity. We expect our studies to be generalizable thereby enabling further regioselective transformations.