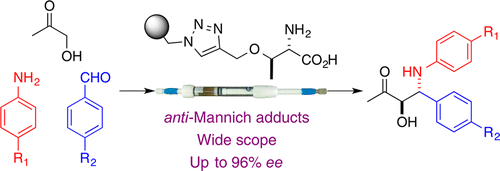
A series of primary amino acid-derived polystyrene-supported organocatalysts was tested in anti-selective Mannich reactions. The polystyrene-immobilized threonine derivative showed the best performance in three-component (hydroxyacetone, anilines, and aldehydes) Mannich reactions to provide anti-β-amino-α-hydroxycarbonyl compounds (11 examples; up to 95% ee), and its use could be extended to dihydroxyacetone and protected hydroxyacetones (7 examples; up to 90% ee). The high activity depicted by the catalyst has allowed its implementation in continuous flow. Under this operation mode, the supported threonine catalyst produces anti-Mannich adducts with generally higher diastereo- and enantioselectivity than in batch. A family of five different enantioenriched anti-Mannich adducts has been sequentially prepared in flow by passing different combinations of anilines and aromatic aldehydes over the same sample of catalyst. This confirms the suitability of this methodology for the rapid access to small libraries of enantioenriched compounds.