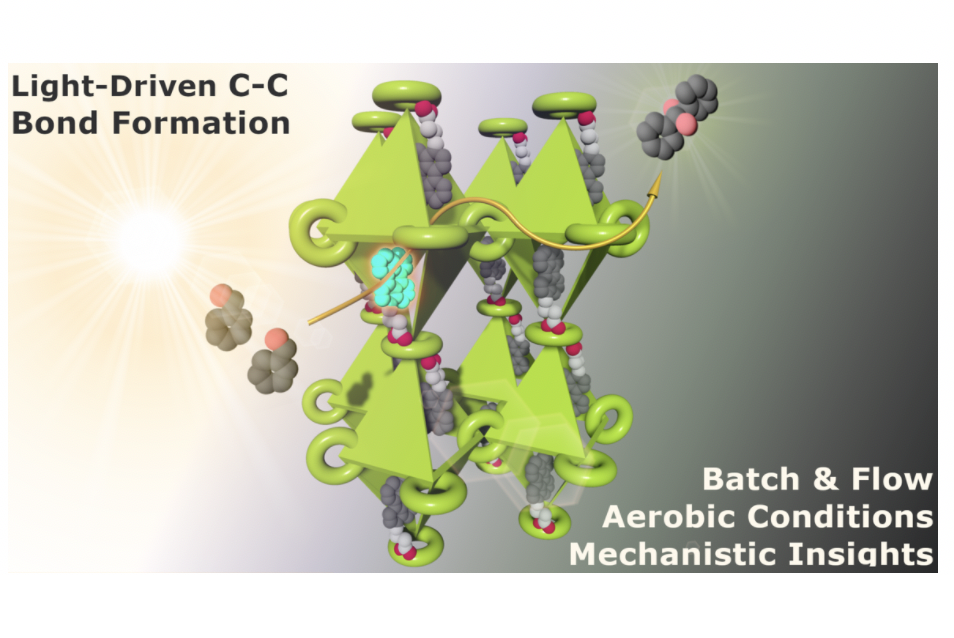
Post‐modification of reticular materials with well‐defined catalysts is an appealing approach to produce new catalytic functional materials with improved stability and recyclability, but also to study the catalysis phenomena in confined spaces. A promising strategy to this end is the post‐functionalization of crystalline and robust metal‐organic frameworks (MOFs) exploiting the potential of crystal‐to‐crystal transformations for further the characterisation of catalysts. In this regard, two new photocatalytic materials, MOF‐520‐PC1 and MOF‐520‐PC2, are straightforwardly obtained by the post‐functionalization of MOF‐520 with perylene‐3‐carboxylic acid (PC1) and perylene‐3‐butyric acid (PC2). The single crystal‐to‐crystal transformation yielded the X‐ray diffraction structure of catalytic MOF‐520‐PC2. The well‐defined disposition of the perylenes inside the MOF served as suitable model systems to get insights into the photophysical properties and mechanism by combining steady‐state, time‐resolved and transient absorption spectroscopy. The resulting materials are active organophotoredox catalysts in the reductive dimerisation of aromatic aldehydes, benzophenones, and imines, under mild reaction conditions. Moreover, MOF‐520‐PC2 can be applied for synthesising gram‐scale quantities of products in continuous‐flow conditions under steady‐state light irradiation. This work provides an alternative approach for the construction of well‐defined metal‐free MOF based catalysts.