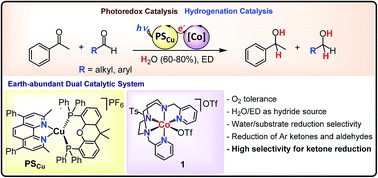
We present an efficient, general, fast, and robust light-driven methodology based on earth-abundant elements to reduce aryl ketones, and both aryl and aliphatic aldehydes (up to 1400 TON). The catalytic system consists of a robust and well-defined aminopyridyl cobalt complex active for photocatalytic water reduction and the [Cu(bathocuproine)(Xantphos)](PF6) photoredox catalyst. The dual cobalt–copper system uses visible light as the driving-force and H2O and an electron donor (Et3N or iPr2EtN) as the hydride source. The catalytic system operates in aqueous mixtures (80–60% water) with high selectivity towards the reduction of organic substrates (>2000) vs. water reduction, and tolerates O2. High selectivity towards the hydrogenation of aryl ketones is observed in the presence of terminal olefins, aliphatic ketones, and alkynes. Remarkably, the catalytic system also shows unique selectivity for the reduction of acetophenone in the presence of aliphatic aldehydes. The catalytic system provides a simple and convenient method to obtain α,β-deuterated alcohols. Both the observed reactivity and the DFT modelling support a common cobalt hydride intermediate. The DFT modelled energy profile for the [Co–H] nucleophilic attack to acetophenone and water rationalises the competence of [CoII–H] to reduce acetophenone in the presence of water. Mechanistic studies suggest alternative mechanisms depending on the redox potential of the substrate. These results show the potential of the water reduction catalyst [Co(OTf)(Py2Tstacn)](OTf) (1), (Py2Tstacn = 1,4-di(picolyl)-7-(p-toluenesulfonyl)-1,4,7-triazacyclononane, OTf = trifluoromethanesulfonate anion) to develop light-driven selective organic transformations and fine solar chemicals.