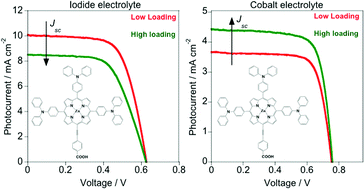
Two zinc–porphyrin sensitizers, 1a and 1b, bearing triphenylamine donor groups, were synthesized and their efficiencies measured in nanocrystalline TiO2 dye sensitized solar cells employing iodide/tri-iodide and tris(1,10-phenanthroline) cobalt electrolytes. Optimized sensitization time for the TiO2 photoanode was found to depend on the electrolyte employed: devices based on iodide/tri-iodide showed better efficiencies with shorter sensitization times (1.5 hours) whereas those based on tris(1,10-phenanthroline) cobalt showed better efficiencies with longer sensitization times (6 hours). From UV-Vis absorption spectra it is estimated that there is roughly twice as much dye loaded onto the TiO2 film sensitized for 6 hours compared to the 1.5 hour film. Interfacial processes were probed using transient photovoltage, transient absorption and fluorescence lifetime measurements. The results indicate that sensitization time does not affect either dye regeneration or interfacial recombination processes in the presence of either electrolyte. However, sensitization time does have a considerable impact on device photocurrent, and moreover, the effect is different for the two electrolytes studied. This work demonstrates how device preparation must be tailored carefully depending on the electrolyte red/ox couple used.