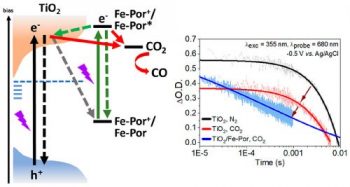
Electro- and photochemical CO2 reduction (CO2R) is the quintessence of modern-day sustainable research. We report our studies on the electro- and photoinduced interfacial charge transfer occurring in a nanocrystalline mesoporous TiO2 film and two TiO2/iron porphyrin hybrid films (meso-aryl- and β-pyrrole-substituted porphyrins, respectively) under CO2R conditions. We used transient absorption spectroscopy (TAS) to demonstrate that, under 355 nm laser excitation and an applied voltage bias (0 to −0.8 V vs Ag/AgCl), the TiO2 film exhibited a diminution in the transient absorption (at −0.5 V by 35%), as well as a reduction of the lifetime of the photogenerated electrons (at −0.5 V by 50%) when the experiments were conducted under a CO2 atmosphere changing from inert N2. The TiO2/iron porphyrin films showed faster charge recombination kinetics, featuring 100-fold faster transient signal decays than that of the TiO2 film. The electro-, photo-, and photoelectrochemical CO2R performance of the TiO2 and TiO2/iron porphyrin films are evaluated within the bias range of −0.5 to −1.8 V vs Ag/AgCl. The bare TiO2 film produced CO and CH4 as well as H2, depending on the applied voltage bias. In contrast, the TiO2/iron porphyrin films showed the exclusive formation of CO (100% selectivity) under identical conditions. During the CO2R, a gain in the overpotential values is obtained under light irradiation conditions. This finding was indicative of a direct transfer of the photogenerated electrons from the film to absorbed CO2 molecules and an observed decrease in the decay of the TAS signals. In the TiO2/iron porphyrin films, we identified the interfacial charge recombination processes between the oxidized iron porphyrin and the electrons of the TiO2 conduction band. These competitive processes are considered to be responsible for the diminution of direct charge transfer between the film and the adsorbed CO2 molecules, explaining the moderate performances of the hybrid films for the CO2R.