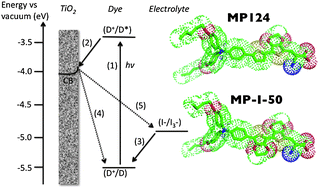
Two new D-π-A type org. sensitizers, MP124 and MP-I-50, were synthesized and their electrochemical and spectroscopic properties studied. Efficiencies of DSSC devices utilizing these dyes were also investigated, where sensitization solvent, sensitization time and additive concentration were all varied. Under standard AM 1.5G simulated solar radiation, optimized MP124 devices show an efficiency of 7.45% (Voc = 0.73 V; Jsc = 14.44 mA cm-2; FF = 70%) while optimized MP-I-50 devices show an efficiency of 5.66% (Voc = 0.68 V; Jsc = 12.06 mA cm-2; FF = 69%). Transient absorption spectroscopy studies show that regeneration of dye cations by the red-ox electrolyte was more efficient in MP124 cells which is attributed to its higher HOMO energy leading to greater driving force for the regeneration reaction. Transient photo voltage studies showed that electron lifetimes were longer lived in MP124 explaining the higher Voc for these cells compared to MP-I-50 cells. DFT and MP2 calculations indicate that this is due to the greater tendency of MP-I-50 to form charge-transfer complexes with the I2 species in the electrolyte, due to the presence of an additional EDOT in its structure compared to MP124. This work highlights the effect that small changes to the sensitizer structure can have on the interfacial charge transfer reactions and ultimately on the device efficiency.