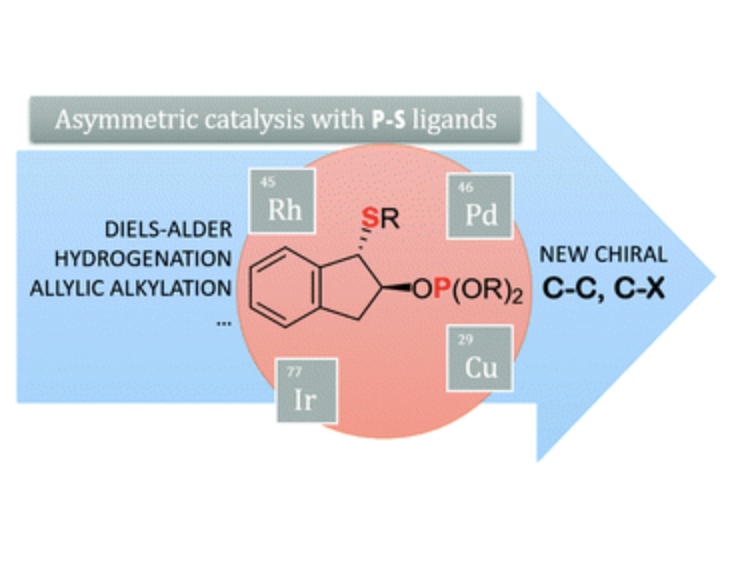
In the early 1990s chiral P–thiother ligands emerged as promising ligands in the field of asymmetric catalysis, with the development of many P–thioether ligand families. However, only a few of them have shown a broad reaction and substrate scope. So, compared with other heterodonor ligands such as the widely studied P–N ligands, their impact in asymmetric catalysis was not realised until recently. This has been mainly attributed to the difficulty of controlling the configuration at the sulfur atom when coordinated to the metal. More recently, it has been found that this problem could be solved by a rigorous choice of the ligand scaffold, a process usually aided by mechanistic studies. This allowed the recent discovery of new P–thioether ligand families with a broader versatility, both in reactions and in substrate/reagent scope. This feature article aims to highlight those new P–thioether ligand libraries and the relationship between the structure and catalytic performance.