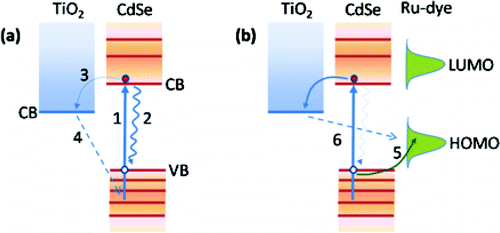
Interacting properties of colloidal CdSe quantum dots (QDs) and polypyridyl ruthenium dyes employed as cosensitizers of mesoporous TiO2 electrodes as well as the effect of QDs coating and anchoring mode (direct and linker adsorption) have been investigated by photoluminesce (PL), Raman, and transient absorption (TAS) spectroscopies. Direct adsorption of QDs on TiO2 leads to a more efficient PL quenching compared to that of QDs attached by means of a mol. linker (cysteine). This fact suggests higher electron injection for the former anchoring mode. Coating of ZnS on CdSe QDs sensitized TiO2 electrodes passivates the QDs surface states and partially releases quantum confinement effects, as is obsd. in colloidal core-shell nanoparticles. Subsequent cosensitization with a ruthenium mol. dye dramatically quenches the PL of the QDs/TiO2 electrodes, even in the presence of ZnS coating, indicating the presence of strong photoinduced charge transfer between the CdSe QDs and the dye mols. This is firmly supported by TAS spectroscopy on the interfacial recombination kinetics that points to the fast hole transfer from the photoexcited QDs to the dye. The regenerating action of mol. dyes for QD sensitizers can have important implications in the development of efficient photovoltaic devices based on the synergistic action of dye-QD-TiO2 heterostructures.