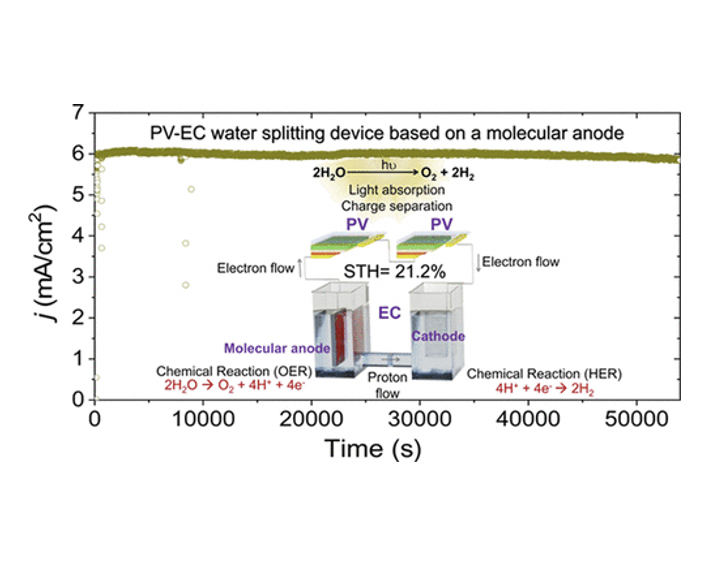
In the urgent quest for green energy vectors, the generation of hydrogen by water splitting with sunlight occupies a preeminent standpoint. The highest solar-to-hydrogen (STH) efficiencies have been achieved with photovoltaic-electrochemical (PV-EC) systems. However, most PV-EC water-splitting devices are required to work at extreme conditions, such as in concentrated solutions of HClO4 or KOH or under highly concentrated solar illumination. In this work, a molecular catalyst-based anode is incorporated for the first time in a PV-EC configuration, achieving an impressive 21.2% STH efficiency at neutral pH. Moreover, as opposed to metal oxide-based anodes, the molecular catalyst-based anode allows us to work with extremely small catalyst loadings (<16 nmol/cm2) due to a well-defined metallic center, which is responsible for the fast catalysis of the reaction in the anodic compartment. This work paves the way for integrating molecular materials in efficient PV-EC water-splitting systems.