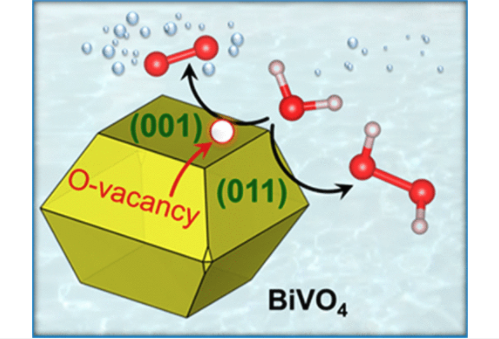
Bismuth vanadate (BiVO4) is one of the most promising photoanode materials for water oxidation. However, the water oxidation mechanism and selectivity on the different surfaces of BiVO4 are still not well understood, partly because of the structural complexity introduced by the presence of oxygen vacancies in the material. Using density functional theory, we show that the (001) surface of BiVO4 with subsurface vacancies is the most suitable for the oxygen evolution reaction, whereas the pristine (011) surface favors the hydrogen peroxide evolution reaction. A mechanism by which the vacancies can be removed from the surface, thereby influencing the water oxidation selectivity, is also described. Our results thus emphasize the crucial impact of the local structure on the catalytic selectivity in ternary oxides.