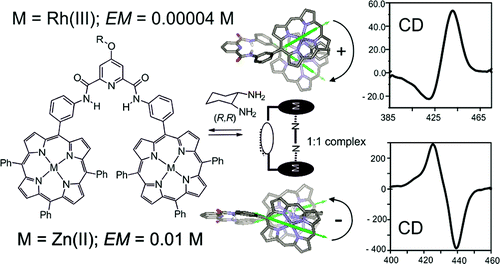
We describe the synthesis of a bisporphyrin tweezer receptor 1•H4 and its metalation with Zn(II) and Rh(III) cations. We report the thermodynamic characterization of the supramolecular chirality induction process that takes place when the metalated bisporphyrin receptors coordinate to enantiopure 1,2-diaminocyclohexane in two different solvents, toluene and dichloromethane. We also performed a thorough study of several simpler systems that were used as models for the thermodynamic characterization of the more complex bisporphyrin systems. The initial complexation of the chiral diamine with the bisporphyrins produces a 1:1 sandwich complex that opens up to yield a simple 1:2 complex in the presence of excess diamine. The CD spectra associated with the 1:1 and 1:2 complexes of both metalloporphyrins, 1•Zn2 and 1•Rh2, display bisignate Cotton effects when the chirogenesis process is studied in toluene solutions. On the contrary, in dichloromethane solutions, only 1•Zn2 yields CD-active 1:1 and 1:2 complexes, while the 1:2 complex of 1•Rh2 is CD-silent. In both solvents, porphyrin 1•Zn2 features a stoichiometrically controlled chirality inversion process, which is the sign of the Cotton effect of the 1:1 complex is opposite to that of the 1:2 complex. In contrast, porphyrin 1•Rh2 affords 1:1 and 1:2 complexes in toluene solutions with the same sign for their CD couplets. Interestingly, in both solvents, the signs of the CD couplets associated with the 1:1 sandwich complexes of 1•Zn2 and 1•Rh2 are opposite. The amplitudes of the CD couplets are higher for 1•Zn2 than for 1•Rh2. This observation is in agreement with 1•Rh2 having a smaller extinction coefficient than 1•Zn2. We performed DFT-based calculations and assigned molecular structures to the 1:1 and 1:2 complexes that explain the observed signs for their CD couplets. Unexpectedly, the quantification of the thermodynamic stability of the two metallobisporphyrin/diamine 1:1 sandwich complexes revealed the existence of interplay between effective molarity values (EM) and the strength of the intermolecular interaction (Km; N•••Zn or N•••Rh) used in their assembly. The EM for the N•••Rh(III) intramolecular interaction is 3 orders of magnitude smaller than that for the N•••Zn(II) interaction, both of which are embedded in the same scaffold of the 1•M2 bisporphyrin receptor.