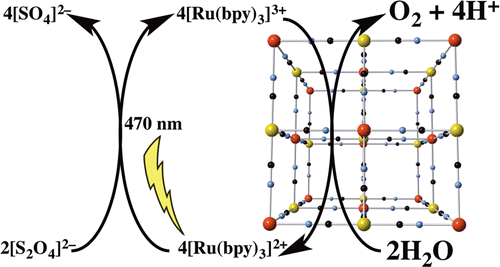
The catalytic activity of metal hexacyanometallates (Prussian blue-type coordination polymers) was tested in photoactivated water oxidation by [Ru(2-2′-bpy)3]2+/persulfate system at pH close to neutrality. This screening shows that manganese and cobalt N-coordinated compounds are able to catalyze the oxygen evolution reaction. Mn hexacyanometalates show low activity. Co hexacyanometalates, in contrast, appear highly active and promote robust catalysis with excellent quantum yield (≤88%), even in acidic media. Our results indicate that these catalysts are viable candidates for a photoactivated water oxidation process as part of an artificial photosynthesis scheme.