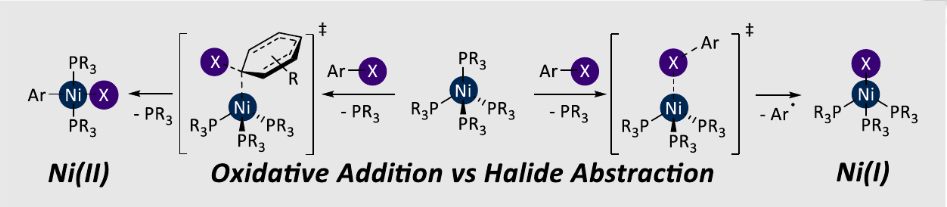
Density functional theory (DFT) calculations have been used to study the oxidative addition of aryl halides to [Ni(PMe3)4]. The formation of NiI versus NiII has been rationalised through the study of two different pathways, both of which proceed via [Ni(PMe3)3]. [NiX(PMe3)3] forms via an open-shell singlet transition state with concommitant formation of an aryl radical, while [Ni(Ar)X(PMe3)2] is produced via SN2-type oxidative addition followed by phosphine dissociation. A microkinetic model was used to show that these data are consistent with the experimentally-observed ratios of NiI and NiII. Importantly, the [Ni(PMe3)2] complex has little if any role in the oxidative addition reaction because it is relatively high in energy. The behaviour of [Ni(PR3)4] complexes in catalysis is likely to differ considerably from those based on diphosphine ligands.