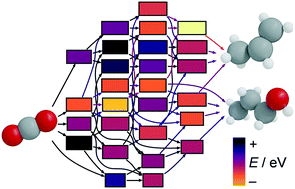
The electrocatalytic CO2 reduction (eCO2R) reaction powered by renewable electricity holds promise for the sustainable production of multi-carbon chemicals and fuels. On Cu-based catalysts, ethylene and ethanol (C2) have been produced in appreciable amounts. C3 products (mostly terminal oxygenates) have limited yields, whereas propylene is puzzlingly absent. Herein, we devise a divide-and-conquer strategy to explain the formation of the C3-backbone and elucidate the mechanism responsible for the observed selectivity by combining network graphs, density functional theory, and experiments to prune the network and benchmark the identified path. Our approach concludes that the most frequently reported products, propionaldehyde and 1-propanol, originate from the coupling of CH2CH with C(H)O. While propylene and 1-propanol share common intermediates, the former is barely produced due to the unfavourable formation of allyl alkoxy (CH2CHCH2O), whose crucial nature was confirmed experimentally. This work paves the way for tailoring selective routes towards C3 products via eCO2R.