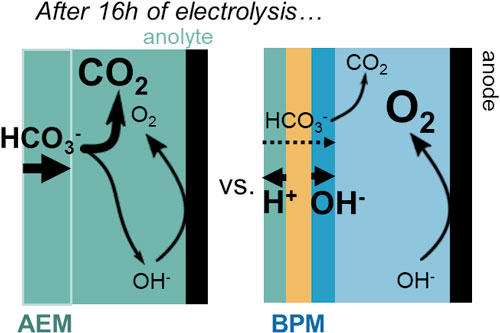
The selectivity of CO2 electrolyzers has hitherto mainly been associated with the cathode selectivity. A few recent studies have shown that the nature of the polymer membrane can impact the system ionic selectivity, with anion exchange membranes (AEM) leading to high crossover of (bi)carbonates during operation and a CO2 pumping effect. In the present work, we investigate and compare CO2 crossover during operation through an AEM and a bipolar membrane (BPM) in a flow cell fed with gaseous CO2. With AEM, starting with 1 M KHCO3 catholyte and 1 M KOH anolyte, the anolyte pH rapidly drops from 14 to 8. This triggers an increase of 1.2 V in cell voltage at 45 mA·cm−2, due to increased OER overpotential and anolyte resistance. Steady-state operation at 45 mA·cm−2 with the AEM results in a CO2/O2 ratio of 3.6 at the anode. With BPM, the anolyte pH decreases more slowly, and the CO2/O2 ratio at the anode under steady-state at 45 mA·cm−2 is only 0.38. Overall, the cell voltage is lower with the BPM than with the AEM at steady-state. These results show the potential of BPMs to mitigate carbon crossover, which could be further reduced by optimizing their design.