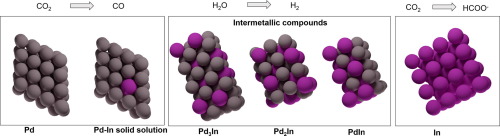
CO2 electroreduction powered by renewable energy is an attractive strategy to close the carbon cycle. Among the possible reduction products, CO is of particular interest due to its large industrial applications. Transition metals in the Pt group are able to electrochemically reduce CO2 to CO, but suffer from CO surface poisoning, which causes a quick deactivation and overall sluggish kinetics. Here, we show that by introducing In to Pd-rich bimetallic particles we can tune the selectivity and limit the surface poisoning of these catalysts. The addition of large amounts of In blocks CO2 reduction activity and leads to a material selective for hydrogen evolution and insensitive to CO poisoning. This study provides insights into the dependence of CO2 reduction selectivity on the composition of Pd-In nanoparticles, revealing the effect that different phases have on catalytic activity. The application of similar screenings to other bimetallic systems can potentially yield cheap, selective, and poison-resistant catalysts for electrochemical applications.