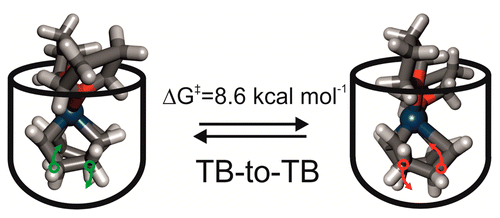
We report experimental and theoretical evidence of restrained axial rotation for heteroleptic L2·IrI·1,5-cyclooctadiene (COD) complexes included in the aromatic cavity of Rebek’s self-folding octaamide cavitand. At 298 K, the axial spinning motion of the included organometallic guests was slow on the 1H NMR time scale and produced a proton spectrum for the bound host indicative ofC2 symmetry. Signals corresponding to aromatic protons of the bound host coalesced at 323 K, indicating that the spinning process of the included guest became fast on the 1H NMR time scale and that the complex approached C4 symmetry. Surprisingly, lowering the temperature of the solution to 193 K induced an additional splitting of the proton signals observed at room temperature for both the bound host and the included guest. We propose the emergence of a new element of chirality in the complexes, which was associated with a slow interconversion, on the 1H NMR time scale between the two chiral twisted-boat conformers of the chelated COD included in the already chiral cavity of the container. This leads to the inclusion complexes existing in solution as pairs of two racemic diastereomers. We estimated that the racemization barrier for the two cyclochiral conformers of the IrI chelated COD was 5 kcal mol–1 higher as an included organometallic complex than as free in solution. Furthermore, we performed a van’t Hoff plot and determined that the inclusion of the organometallic complex in the cavitand was endothermic and exclusively driven by entropy (ΔH = 5.9 kcal mol–1 and ΔS = 33.9 cal mol–1 K–1).