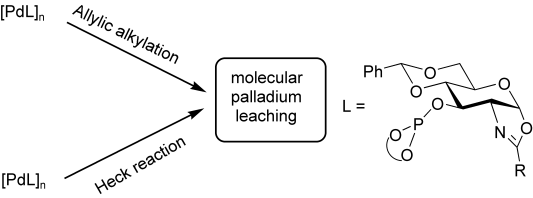
A series of palladium nanoparticles stabilized by five chiral sugar-based oxazolinyl-phosphite ligands, containing various substituents at the oxazoline and phosphite moieties has been synthesized. They were characterized by transmission electron microscopy (TEM), X-ray powder diffraction (XRD), infrared spectroscopy (IR) and elemental analysis. These nanoparticles were applied in Pd-catalyzed asymmetric allylic alkylation and Heck coupling reactions. A detailed study to elucidate the nature of the active species using a continuous-flow membrane reactor (CFMR), accompanied by TEM observations, classical poisoning experiments, and kinetic measurements have been carried out. Conclusive evidence of the nature of the species involved in the use of PdNPs in asymmetric catalytic reactions has been obtained. The CFMR experiments proved the molecular nature of the true catalysts and all conversions can be justified by the amount of molecular palladium that leached as measured by ICP-AES.