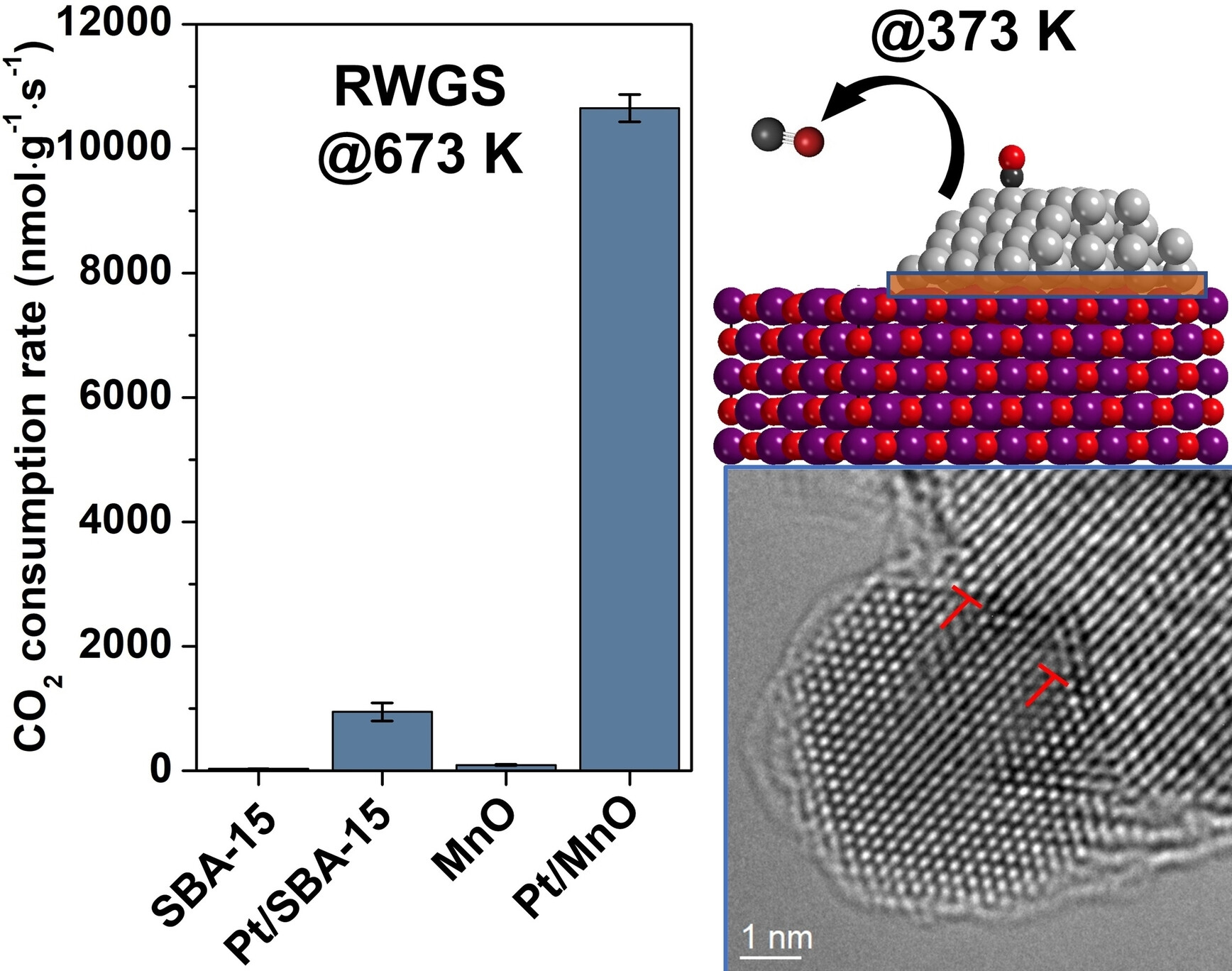
The implementation of supported metal catalysts heavily relies on the synergistic interactions between metal nanoparticles and the material they are dispersed on. It is clear that interfacial perimeter sites have outstanding skills for turning catalytic reactions over, however, high activity and selectivity of the designed interface-induced metal distortion can also obtain catalysts for the most crucial industrial processes as evidenced in this paper. Herein, the beneficial synergy established between designed Pt nanoparticles and MnO in the course of the reverse water gas shift (RWGS) reaction resulted in a Pt/MnO catalyst having ≈10 times higher activity compared to the reference Pt/SBA-15 catalyst with >99 % CO selectivity. Under activation, a crystal assembly through the metallic Pt (110) and MnO evolved, where the plane distance differences caused a mismatched-row structure in softer Pt nanoparticles, which was identified by microscopic and surface-sensitive spectroscopic characterizations combined with density functional theory simulations. The generated edge dislocations caused the Pt lattice expansion which led to the weakening of the Pt−CO bond. Even though MnO also exhibited an adverse effect on Pt by lowering the number of exposed metal sites, rapid desorption of the linearly adsorbed CO species governed the performance of the Pt/MnO in the RWGS.