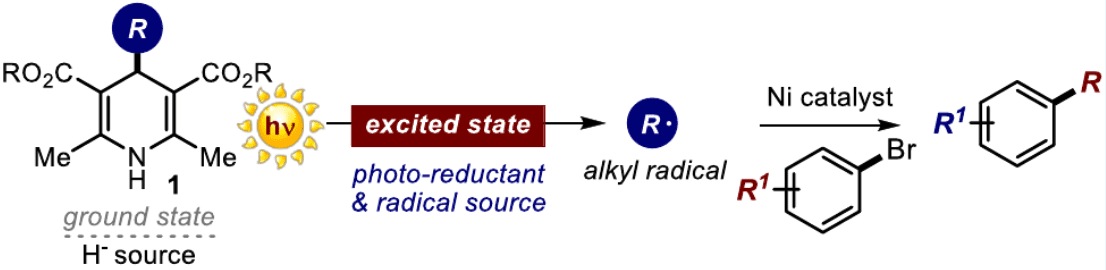
We report herein that 4-alkyl-1,4-dihydropyridines (alkyl-DHPs) can directly reach an electronically excited state upon light absorption and trigger the generation of C(sp3)-centered radicals without the need for an external photocatalyst. Selective excitation with a violet-light-emitting diode turns alkyl-DHPs into strong reducing agents that can activate reagents via single-electron transfer manifolds while undergoing a homolytic cleavage to generate radicals. We used this photochemical dual-reactivity profile to trigger radical-based carbon-carbon bond-forming processes, including nickel catalyzed cross-coupling reactions.